


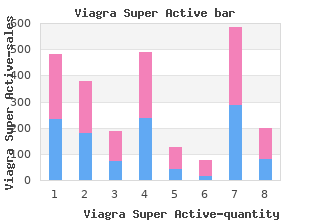
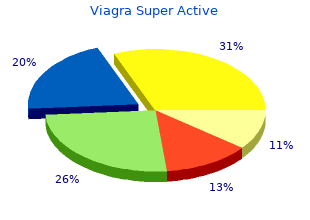
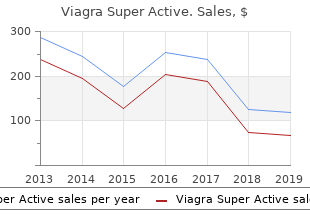
Northwest Missouri State University. C. Angar, MD: "Purchase Viagra Super Active - Discount Viagra Super Active online OTC".
Participants consistently attributed this poor confdence to unplanned pregnancies following a 1998 lapse in the quality of oral contraceptives (Associated Press purchase viagra super active 25 mg free shipping erectile dysfunction caused by hydrocodone, 1998; Goering cheap viagra super active 50mg visa erectile dysfunction melanoma, 1998) order viagra super active 50 mg without a prescription erectile dysfunction caused by anabolic steroids. Anvisa, the Brazilian drugs regulatory authority, was created in response to this and other medicine quality problems (Csillag, 1998). They are evidence, however, that fake medicine can do long-term damage to the reputation of the health system. Social and Developmental Costs In a larger sense, trade in falsifed and substandard medicines under- mines not just the health system but all public institutions. Falsifed medicines are often the business of criminal car- tels, including the Camorra crime group in Naples, the Russian mafa, and Latin American drug cartels, and terrorist organizations, such as Al-Qaeda and Hezbollah (Findlay, 2011). Fake medicines generate income for criminals, and only the weakest evidence, if any, ties them to their crime. Acute cases of medicine poisoning can elicit public outcry, but more often bad drugs go unnoticed, blending in with lawful business. Victims of falsifed and substandard drugs usually do not even know they are victims and are therefore deprived of their right to redress. In many parts of the world, falsifed and substandard medicines further erode the already weak political infrastructure that allows them to circulate, part of a vicious cycle of poverty and crime. Countering the Problem of Falsified and Substandard Drugs 75 Copyright © National Academy of Sciences. Countering the Problem of Falsified and Substandard Drugs 76 Copyright © National Academy of Sciences. Countering the Problem of Falsified and Substandard Drugs 77 Copyright © National Academy of Sciences. Diagnosing renal failure due to diethylene glycol in children in a resource-constrained setting. High heritability of malaria parasite clearance rate indicates a genetic basis for artemisinin resistance in Western Cambodia. Distribution of causes of maternal mortality among different socio-demographic groups in Ghana: A descriptive study. Quality of private and public ambu- latory health care in low and middle income countries: Systematic review of comparative studies. False resistance to antiparasitic drugs: Causes from shelf availability to patient compliance. Medicines prices, availability, and affordability in 36 developing and middle-income countries: A secondary analysis. Fatal poisoning among young children from diethylene glycol-contaminated acetaminophen. In vitro assessment of qual- ity control parameters of some commercially available generics of amlodipine besylate in Nigerian drug market. A 6-year (2004-2009) review of maternal mortality at the eastern regional hospital, Koforidua, Ghana. Vaccine supply chains need to be better funded and strengthened, or lives will be at risk. Epidemic of plasmodium falciparum malaria involving substandard antimalarial drugs, Pakistan, 2003. Are counterfeit or substandard anti-infective products the cause of treatment failure in Papua New Guinea? Institute of legal medicine advances in review of pending cases: 219 are killed by syrup. Burden of disease caused by streptococcus pneumoniae in children younger than 5 years: Global estimates. Comparative assessment of the qual- ity control measurements of multisource amlodipine tablets marketed in Nigeria. Epidemiological survey of mithicillin resistant Staphylococcus aureus in the community and hospital, Gannavaram, Andhra Pradesh, South India. Emergence of artemisinin-resistant malaria on the western border of Thailand: A longitudinal study. Antibiotic use, re- sistance development and environmental factors: A qualitative study among healthcare professionals in Orissa, India. Uterotonic drug quality: An assessment of the potency of injectable uterotonic drugs purchased by simulated clients in three districts in Ghana. A cluster of cases of severe cardiotoxicity among kala- azar patients treated with a high-osmolarity lot of sodium antimony gluconate. Mitigating the threat of artemisinin resistance in Africa: Improvement of drug-resistance surveillance and response systems. Infuence of tropical climate conditions on the quality of antihypertensive drugs from Rwandan pharmacies. International study of the prevalence and outcomes of infection in intensive care units. Hyperparasitaemia and low dosing are an important source of anti-malarial drug resistance. Survey of the quality of antiretroviral medicines circulating in selected African countries. Counterfeit and substandard drugs in Myanmar and Viet Nam: Report of a study carried out in cooperation with the governments of Myanmar and Viet Nam. Countering the Problem of Falsified and Substandard Drugs Copyright © National Academy of Sciences. Countering the Problem of Falsified and Substandard Drugs 3 The Magnitude of the Problem It is diffcult to accurately measure the burden of falsifed and sub- standard drugs. As Chapter 2 mentions, some of the understanding of the problem comes from alerts in gray literature, including investigative journalism and industry and association reporting. Although these sources provide some insight, they do not provide an accurate estimate of the true magnitude of the problem. National regulatory authorities and drug com- panies keep records on fraudulent medicines; a broader understanding of the problem comes from peer-reviewed literature. There are few epidemio- logically rigorous, peer-reviewed studies on the prevalence of falsifed and substandard drugs. This chapter presents the results of a cross section of government and industry data and peer-reviewed and gray literature about the global burden of falsifed and substandard drugs. It does not summarize every study, but rather gives an overview of important trends. There was a time when this was a con- scious secrecy, an effort to avoid discrediting the public health system and drug companies, expressed in the Royal Pharmaceutical Society’s 1989 statement “no great publicity [about fake drugs] should be sought because it could damage public confdence in medicines” (Cockburn et al. Food and Drug Administration and Interpol, have taken action against sub- standard and falsifed medicines. Similarly, drug companies, both innovator and generic, may withhold information about falsifed and substandard medicines on the grounds that such stories discourage patient confdence in their products (Cockburn et al.
Hydroxocobalamin is the form of vitamin B12 used to treat vitamin B12 defciency due to dietary defciency or malabsorp- ton (see chapter 13 buy viagra super active discount erectile dysfunction treatment in kuala lumpur. Folic acid should not be used in undiagnosed megaloblastc anaemia unless vitamin B12 is administered concurrently order 100mg viagra super active mastercard erectile dysfunction yahoo, otherwise neuropathy may be precipitated (see chapter 13 best purchase viagra super active erectile dysfunction without pills. Supplementaton with folic acid 500 µg daily is recommended for women of child-bearing potental in order to reduce the risk of serious neural tube defects in their ofspring. Claims that ascorbic acid is of value in the treatment of common colds are unsubstantated. The term vitamin D covers a range of compounds including ergocalciferol (vitamin D2) and cholecalciferol (vitamin D3). These two compounds are equipotent and either can be used to prevent and treat rickets. Minerals: Calcium gluconate: Calcium supplements are usually only required where dietary calcium intake is defcient. This dietary requirement varies with age and is relatvely greater in child- hood, pregnancy and lactaton due to an increased demand, and in old age, due to impaired absorpton. In osteoporosis, a calcium intake which is double the recommended daily amount reduces the rate of bone loss. In hypocalcaemic tetany calcium gluconate must be given parenterally but plasma calcium must be monitored. The recommended intake of iodine is 150 µg daily (200 µg daily in pregnant and lactaton women); in children the recommended intake of iodine is 50 µg daily for infants under 1 year, 90 µg daily for children aged 2-6 years, and 120 µg daily for children aged 7-12 years. Defciency causes endemic goitre and results in endemic cretnism (characterized by deaf-mutsm, intellectual defcit, spastcity and sometmes hypothyroidism), impaired mental functon in children and adults and an increased inci- dence of stll-births and perinatal and infant mortality. Iodine and iodides may suppress neonatal thyroid functon and in general iodine compounds should be avoided in pregnancy. Where it is essental to prevent neonatal goitre and cretnism, iodine should not be witheld from pregnant women. Control of iodine defciency largely depends upon salt iodizaton with potassium iodide or potassium iodate and through dietary diversifcaton. In areas where iodine defciency disorders are moderate to severe, iodized oil given either before or at any stage of pregnancy is found to be benefcial. Sodium fuoride: Availability of adequate fuoride confers signifcant resistance to dental caries. It is now considered that the topical acton of fuoride on enamel and plaque is more important than the systemic efect. Where the natural fuoride content of the drinking water is signifcantly less than 1 mg per litre, artfcial fuoridaton is the most economical method of supplementng fuoride intake. Daily administra- ton of fuoride tablets or drops is a suitable alternatve, but systemic fuoride supplements should not be prescribed without reference to the fuoride content of the local water supply; they are not advisable when the water contains more than 700 µg per litre. Individuals who are either partcularly caries prone or medically compromized may be given additonal protecton by the use of fuoride rinses or by applicaton of fuoride gels. Rinses may be used daily or weekly; daily use of a less concen- trated rinse is more efectve than weekly use of a more concentrated one. High-strength gels must be applied on a regular basis under professional supervision; extreme cauton is necessary to prevent the child from swallowing any excess. Ascorbic Acid (Vitamin C)* Pregnancy Category-A, C Indicatons Preventon and treatment of scurvy. Adverse Efects Gastrointestnal disturbances reported with large doses; failure of concepton; kidney oxalate stones. Calcium Carbonate + Vitamin D3 Pregnancy Category-A Indicatons Preventon and treatment of osteoporosis and osteomalacia, nutritonal supplement. Contraindicatons Hypersensitvity to any of the components, hypercalcaemia and/or hypercalciuria, nephrolithiasis, hypervitaminosis. Precautons Renal impairment, impaired calcium absorpton in achlorhydria, risk of hypercalcaemia and hypercalciuria in hypoparathyroid patents receiving high doses of vitamin D; interactons (Appendix 6c); pregnancy (Appendix 7c). Adverse Efects Constpaton, fatulence, nausea, abdominal pain and diarrhoea; pruritus, rash and urtcaria. Dose Slow intravenous injecton and contnuous intravenous infusion Adult- Hypocalcaemic tetany: 1g (2. Contraindicatons Conditons associated with hypercalcaemia and hypercalciuria (for example some forms of malignant disease). Precautons Monitor plasma calcium concentraton; renal impairment; interactons (Appendix 6c); diarrhoea, parathyroid disease; stomach trouble. Adverse Efects Mild gastrointestnal disturbances; bradycardia, arrhythmias, hypotension; irritaton at injecton site; sof tssue calcifcaton; nephrocalcinosis, renal calculi. Ergocalciferol (Vitamin D2)* Pregnancy Category-C Indicatons Preventon of vitamin D defciency; vitamin D defciency caused by malabsorpton or chron- ic liver disease; hypocalcaemia of hypopar- athyroidism; osteomalacia; osteoporosis. Dose Oral Adult and child- Preventon of vitamin D defciency: 10 µg (400 units) daily. Precautons Ensure correct dose in infants; monitor plasma calcium at weekly intervals in patients receiving high doses or those with renal impairment; nausea and vomiting- may indicate overdose and hypercalcaemia; lactation (Appendix 7b); interactions (Appendix 6a); pregnancy (Appendix 7c). Adverse Efects Symptoms of overdosage include anorexia; lassitude; nausea and vomitng, diarrhoea, weight loss; polyuria; sweatng; headache; thirst, vertgo and raised concentratons of calcium and phosphate in plasma and urine; tssue calcifcaton may occur if dose of 1. Dose Oral Adult- Endemic moderate to severe iodine defciency: during pregnancy and one year postpartum, 300 to 480 mg once a year or 100 to 300 mg every 6 months; women of child-bearing age, 400 to 960 mg once a year or 200 to 480 mg every 6 months. Child- Iodine defciency: infant under 1 year, single dose 100 mg; 1 to 5 years, 200 mg once a year; above 6 years 400 mg once a year. Contraindicatons Lactaton (Appendix 7b); bronchits; goitre; hyperkalaemia; asthma; acne vulgaris; tuberculosis. Precautons Over 45 years old or with nodular goitre (especially susceptble to hyperthyroidism when given iodine supplements-iodized oil may not be appropriate); may interfere with thyroid-functon tests; pregnancy (see notes above and Appendix 7c); acute iodide toxicity; cardiac toxicity; interactons (Appendix 6c). Adverse Efects Hypersensitvity reactons; goitre and hypothyroidism; hyperthyroidism; bronchits; eosinophilia; rashes; headache; salivaton. Dose Initally 1000 µg 3 tmes a day for 2 weeks, thereafer 1000 µg every 3 months by intramuscular injecton in case of pernicious anaemia and other macrocytc anaemia. Nicotnamide* Pregnancy Category-A Indicatons Treatment of pellagra; hartnup disease; infammatory skin disease. Precautons Avoid contact with eyes and mucous membranes (including nose and mouth); reduce frequency of applicaton if excessive dryness, irritaton or peeling; history of heart disease; insulin dependent diabetes; pregnancy (Appendix 7c). Adverse Efects Dryness of skin; also pruritus, erythema, burning and irritaton; hepatotoxicity, cholestasis; portal fbrosis; transient liver dysfuncton; tautness of face. Ribofavin* Pregnancy Category-A Indicatons Vitamin B2 defciency; arabinofavinosis. Dose Oral Adult and child- Treatment of vitamin B2 defciency: up to 30 mg daily in divided doses. Precautons Large doses result in dark yellow discolouraton of urine; pregnancy (Appendix 7c).
Guidelines for the psychosocially assisted pharmacological treatment of opioid dependence cheap viagra super active online amex erectile dysfunction quotes. European Journal of Clinical Microbiology and Infectious Diseases order viagra super active cheap online erectile dysfunction kamagra, 2012 discount viagra super active amex intracavernosal injections erectile dysfunction, 31:919–927. Scaling up antiretroviral therapy in resource-limited settings: adapting guidance and meet the challenges. Initiating highly active antiretroviral therapy in sub-Saharan Africa: an assessment of the revised World Health Organization scaling-up guidelines. Immunodeficiency at start of combination antiretroviral therapy in low, middle and high income countries. Effect of highly active antiretroviral therapy on incidence of tuberculosis in South Africa: a cohort study. Proceedings of the National Academy of Sciences of the United States of America, 2010, 107:19485–19489. Antiretroviral Pregnancy Registry international interim report for 1 January 1989 through 31 July 2012. Liver-related deaths in persons infected with the human immunodeficiency virus: the D: A:D study. Liver fibrosis progression in human immunodeficiency virus and hepatitis C virus coinfected patients. Impact of human immunodeficiency virus infection on the course of hepatitis C virus infection: a meta- analysis. Minneaolis, Clinical and Translational Science Institute, University of Minnesota, 2012 (http://apps. Adherence to antiretroviral therapy during and after pregnancy in low-income, middle-income, and high- income countries: a systematic review and meta-analysis. Cross Continents Collaboration for Kids (3Cs4kids) Analysis and Writing Committee. Pretoria, Republic of South Africa National Department of Health 2013 (http://www. Adverse events associated with nevirapine and efavirenz-based first-line antiretroviral therapy: a systematic review and meta-analysis. Safety of efavirenz in the first trimester of pregnancy: an updated systematic review and meta- analysis. Technical update on treatment optimization: pharmacological equivalence and clinical interchangeability between lamivudine and emtricitabine, a review of current literature. Adverse events associated with nevirapine use in pregnancy: a systematic review and meta-analysis. Adverse events associated with nevirapine use in pregnancy: a systematic review and meta-analysis. A trial of shortened zidovudine regimens to prevent mother-to-child transmission of human immunodeficiency virus type 1. Switching children previously exposed to nevirapine to nevirapine-based treatment after initial suppression with a protease-inhibitor-based regimen: long-term follow-up of a randomised, open-label trial. Kaletra (lopinavir/ritonavir): label change – serious health problems in premature babies. Reverse transcriptase genotypes in pediatric patients failing initial antiretroviral therapy in Gaborone, Botswana. Simplification of antiretroviral therapy with tenofovir-emtricitabine or abacavir-lamivudine: a randomized, 96-week trial. Conservation of first-line antiretroviral treatment regimen where therapeutic options are limited. Validating clinical and immunological definitions of antiretroviral treatment failure in Malawi. Evaluation of World Health Organization criteria for antiretroviral treatment failure in resource-limited settings. Evaluating patients for second-line antiretroviral therapy in India: the role of targeted viral load testing. Failure of immunologic criteria to appropriately identify antiretroviral treatment failure in Uganda. The role of targeted viral load testing in diagnosing virological failure in children on antiretroviral therapy with immunological failure. Dried blood spots perform well in viral load monitoring of patients who receive antiretroviral treatment in rural Tanzania. Guidelines for the psychosocially assisted pharmacological treatment of opioid dependence. Effect of concomitantly administered rifampin on the pharmacokinetics and safety of atazanavir administered twice daily. Effect of rifampin on steady-state pharmacokinetics of atazanavir with ritonavir in healthy volunteers. Pharmacokinetics of adjusted-dose lopinavir-ritonavir combined with rifampin in healthy volunteers. Resistance in pediatric patients experiencing virologic failure with first- and second-line antiretroviral therapy. Treatment outcomes of patients on second-line antiretroviral therapy in resource-limited settings: a systematic review and meta-analysis. Cardiovascular risk factors in adult Malawians on long-term antiretroviral therapy. Transactions of the Royal Society of Tropical Medicine and Hygiene, 2011, 105:644–649. Length/ height-for-age, weight-for-age, weight-for-length, weight-for-height and body mass index-for-age. Adherence to medication regimens among children with human immunodeficiency virus infection. Adherence to antiretroviral therapy during and after pregnancy in low-income, middle-income, and high- income countries: a systematic review and meta-analysis. Depression, alcohol use and adherence to antiretroviral therapy in sub-saharan Africa: a systematic review. Interventions to increase antiretroviral adherence in sub-Saharan Africa: a systematic review of evaluation studies. Distribution of antiretroviral treatment through self-forming groups of patients in Tete Province, Mozambique. Ambassadors for adherence: provision of highly effective defaulter tracing and re-engagement by peer educators in Tanzania. Effectiveness of collaborative care for depression in human immunodeficiency virus clinics. A pilot study of food supplementation to improve adherence to antiretroviral therapy among food-insecure adults in Lusaka, Zambia. Challenges in using mobile phones for collection of antiretroviral therapy adherence data in a resource- limited setting. Supporting patient adherence to antiretrovirals using mobile phone reminders: patient responses from South India.
Syndromes
Novel electrical detection of label-free disease marker proteins using piezoresistive self-sensing micro-cantilevers buy viagra super active impotence natural treatments. Tracking metastatic tumor cell extravasation with quantum dot nanocrystals and fluorescence emission-scanning microscopy purchase generic viagra super active canada erectile dysfunction treatment in kerala. Superparamagnetic nanoparticles for biomed- ical applications: Possibilities and limitations of a new drug delivery system generic 25mg viagra super active overnight delivery erectile dysfunction pills buy. Polymeric micelles for delivery of poorly soluble drugs: Preparation and anticancer activity in vitro of paclitaxel incorporated into mixed micelles based on poly(ethylene glycol)-lipid conjugate and positively charged lipids. Selective targeting of antibody-conjugated nanoparticles to leukemic cells and primary T-lymphocytes. Nano-encapsulation of azole antifungals: Potential applications to improve oral drug delivery. Synergistic enhancement of selective nanophotothermolysis with gold nanoclusters: Potential cancer therapy. Innovative drug delivery nanosystems improve the anti-tumor activity in vitro and in vivo of anti-estrogens in human breast cancer and multiple myeloma. Liposomal glucocorticoids as tumor- targeted anti-angiogenic nanomedicine in B16 melanoma-bearing mice. Synthesis, characterization and targeting of biodegradable magnetic nanocomposite particles by external magnetic fields. Interaction of functionalized super- paramagnetic iron oxide nanoparticles with brain structures. Lactoferrin and ceruloplasmin derivatized superparamag- netic iron oxide nanoparticles for targeting cell surface receptors. Sub-cellular accumulation of magnetic nanoparti- cles in breast tumors and metastases. Preparation of carbon-coated magnetic iron nanoparticles from composite rods made from coal and iron powders. Plasma synthesis of carbon magnetic nanoparticles and immobilization of doxorubicin for targeted drug delivery. Characterization and anticancer activity of the micelle-forming polymeric anticancer drug Adriamycin-conjugated poly(ethylene glycol)-poly(aspartic acid) block copolymer. Ceramic-based nanoparticles entrapping water-insoluble photosensitizing anticancer drugs: A novel drug-carrier system for pho- todynamic therapy. The embedding of meta-tetra(hydroxyphenyl)chlorin into silica nanoparticle platforms for photodynamic therapy and their singlet oxygen production and pH-dependent optical properties. Influence of particle size on transport of methotrexate across blood brain barrier by polysorbate 80-coated polybutylcyanoacrylate nanoparticles. Encapsulation of 9-nitrocamptothecin, a novel anticancer drug, in biodegradable nanoparticles: Factorial design, characterization and release kinetics. Trastuzumab-modified nanoparticles: Optimisation of preparation and uptake in cancer cells. Poly(ethylene oxide)-modified poly(epsilon-caprolactone) nanoparticles for targeted delivery of tamoxifen in breast cancer. Biodistribution of colloidal gold nanoparticles after intravenous administration: Effect of particle size. Nanomedicine for drug delivery and imaging: A promising avenue for cancer therapy and diagnosis using targeted functional nanopar- ticles. Perumal Department of Pharmaceutical Sciences, South Dakota State University, Brookings, South Dakota, U. Kaushik Department of Biology & Microbiology/Veterinary Science, South Dakota State University, Brookings, South Dakota, U. The biodegradation of proteins into sim- ple amino acids makes them attractive polymers for drug delivery applications. As a result, protein polymers are being increasingly investigated for various nano- enabled drug delivery systems (1). In fact, the first protein-based nanopartic- ulate system is already in the market. In general, nanosystems used in drug delivery range in size from 100 to 1000 nm and are prepared using natural or synthetic polymers or lipids. The drug is encapsulated inside the nanosystem (nanocapsule or nanospheres) or is entrapped in the matrix of the nanosystem (nanoparticles). Alternatively, drugs or other agents of interest are adsorbed, complexed, or conjugated to the surface of the nanosystems. A distinct advantage over syn- thetic polymers is the proven biocompatibility of proteins. Furthermore, they are biodegradable and are broken down into nontoxic by-products. Protein polymers are derived from animals or plants and are, therefore, devoid of monomers or ini- tiators that are found in synthetic polymers. However, the composition and purity of protein biopolymers are difficult to control. Similarly, it is important to protect the protein from premature proteolytic degradation in the body. Although most of the proteins are generally safe, nonautologus proteins can be immunogenic (2). Proteins can be classified into different types based on their source or struc- tural features. This chapter is mainly focused on natural protein polymers derived from animal or plant sources. The properties of different protein polymers used in drug delivery are listed in Table 1. Majority of the published literature on pro- tein nanoparticles is on animal proteins such as gelatin and albumin. On the other hand, there are limited reports on plant protein–based nanoparticles. Animal proteins carry the risk of infection from pathogen contamination with animal tissues. However, animal proteins, such as gelatin, can be easily sterilized due to their thermal stability. In contrast, plant pro- teins are devoid of these issues, and further plant proteins are universally accepted as they can be used even by people who do not consume animal proteins for per- sonal or religious reasons. This chapter discusses the properties of selected animal and plant proteins with an emphasis on factors that influence the preparation of nanoparticles, methods for preparing protein nanoparticles, biological interactions of protein-based nanoparticles, and their drug delivery applications. Gelatins obtained by these two processes differ in their isoelectric point (pI), molecular weight, amino acid composition, and viscosity (6). For example, type A gelatin has a pI of 7 to 9, whereas type B gelatin has a pI of 4 to 5. Apart from its biomedical applications, gelatin is also widely used in the food industry as a stabilizer and a protective coating material.
Cheap 50 mg viagra super active otc. 6 Natural Treatments for Erectile Dysfunction.